Transformative
Medicine
The Miller School’s pioneering role in novel stem cell therapies
By Josh Baxt
Photo illustrations by Ann Cutting
Illustrations by Mike Sudal

Medical science teaches that everybody on Earth began as a single cell. Over the next nine months, that one cell rapidly multiplied and differentiated into more than 200 cell types, and many unique and specialized tissues: skin, brain, liver, heart. Then, when the body was completely formed, the program dramatically slowed down, leaving only a few residual tissue precursor cells in place. Though people retain some regenerative capacity — small reservoirs of cells that can replace damaged tissue — that initial rush of creation, for the most part, never returns. But what if it could? That’s the question being asked by investigators at a variety of research centers throughout the Miller School of Medicine.
Working together to advance this promising field, molecular and cell biologists, neurologists, cardiologists, geriatricians, immunologists, endocrinologists, surgeons and other specialists are making the University of Miami’s Interdisciplinary Stem Cell Institute (ISCI) a leader in stem cell research, both in the lab and through many of the world’s most robust clinical trials. From basic science concepts to leading several clinical trials, ISCI is one of the few centers in the nation that is demonstrating the complete bench-to-bedside approach in this exciting field, testing stem cell treatments for cardiovascular disease, Alzheimer’s, stroke, depression, diabetes, Crohn’s disease, pulmonary fibrosis and many other conditions. If these scientists can harness stem cells to repair or replace diseased tissues, they could solve many of the world’s health problems. The potential is tantalizing.
But there’s a catch. To succeed, scientists must replicate the complex array of molecular signals that tell a pluripotent stem cell to become a neuron rather than a heart muscle cell, for instance.
“We believe one very promising approach to repairing the heart and the brain is not to engraft new cells but to stimulate the organ to regenerate itself.”
— Joshua M. Hare, M.D.
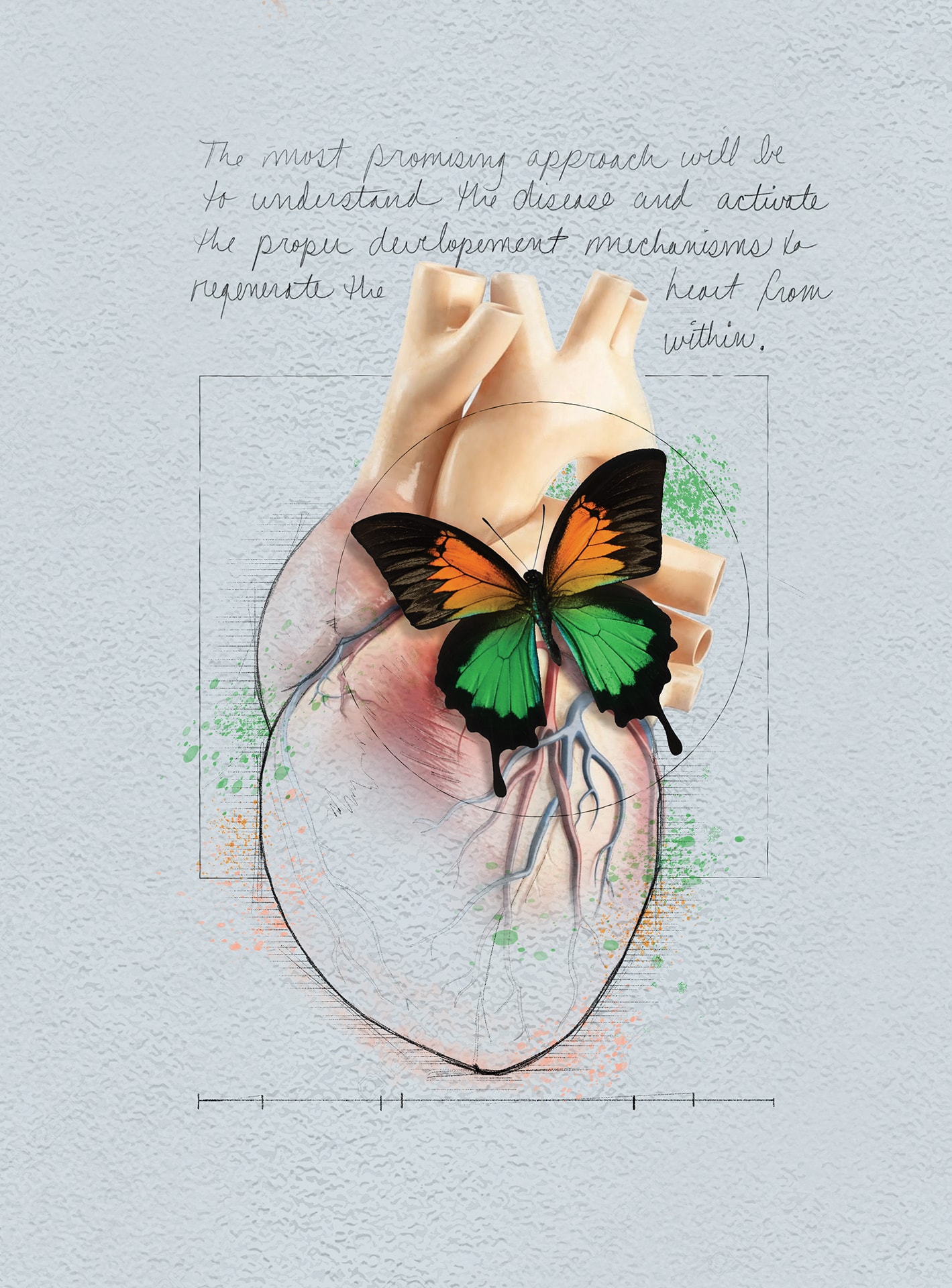
HEALING HEARTS
Cell therapies could take different shapes. There’s the straight regenerative approach of creating tissue to replace skin, neurons, heart muscle or pancreatic beta cells. Heart failure patients could receive replacement muscle to reinvigorate their ailing pumps.
Another approach would stimulate the body’s existing regenerative capacity. Every cell has regenerative genes that have been turned off since birth. If those could be boosted, in a controlled way, they could create new tissue.
“We believe one very promising approach to repairing the heart and the brain is not to engraft new cells but to stimulate the organ to regenerate itself,” said Joshua M. Hare, M.D., founding director of the Miller School’s Interdisciplinary Stem Cell Institute and Louis Lemberg Professor of Medicine.
To pursue this strategy, ISCI scientists have collaborated broadly with colleagues at UM and other institutions across the country to conduct several clinical trials using mesenchymal stem cells and cardiac precursor cells to repair heart damage in patients with a variety of conditions. They also have collaborated with the National Institutes of Health Cell Therapy Research Network on a recently completed study testing a new idea that mixing different cell types might enhance cardiac repair and recovery.
ISCI scientists are leaving no stone unturned, rigorously looking at both approaches. Konstantinos Chatzistergos, Ph.D., research assistant professor of cell biology, has spent years looking to embryonic development for clues.
“We’re trying to understand how and why most of the heart muscle cells are generated during embryogenesis, what happens to those molecular and cellular mechanisms after birth, and how we can reactivate them for regenerative purposes,” he said.
Understanding these processes is crucial. If researchers and clinicians cannot tell cells precisely what to do, attempted therapies could be useless, or worse.
“For example, it is now possible to recapitulate some aspects of human heart development in a dish and produce large amounts of heart muscle cells from stem cells,” Dr. Chatzistergos said. “But these stem cell-derived heart cells are mixtures of right ventricular myocytes, left ventricular myocytes, atrial myocytes and pacemaker cells. If a culture has a high number of pacemaker cells, it can cause arrhythmias. Those arrhythmias led to major limitations on using pluripotent stem cells for heart repair in early studies.”
He and his colleagues have successfully used gene editing to prevent pacemaker cells from forming, setting the stage for therapies to replace damaged heart tissue. But again, the ultimate goal is to activate the heart’s innate regenerative processes.
“The most promising approach will be to understand the disease and activate the proper developmental mechanisms to regenerate the heart from within,” Dr. Chatzistergos said.
Ivonne Schulman, M.D. ’97, professor of clinical medicine, has overseen several efforts to use mesenchymal stem cells to help prevent cardiovascular disease in patients with metabolic syndrome or diabetes. Too much blood glucose damages blood vessel linings, called endothelium, contributing to widespread organ damage.
In two phase 1 and 2 safety trials, Dr. Schulman and her colleagues are infusing mesenchymal stem cells to determine if they can repair or prevent that damage.
“Cardiovascular disease is the No. 1 cause of death in diabetics,” she said. “The mesenchymal stem cells release certain factors that promote vascular repair and vascular health. We want to see if they improve endothelial function. So far, they’ve been quite safe.”
Aisha Khan, M.Sc., M.B.A., executive director of the Clinical Research Cell Manufacturing Program at ISCI, has overseen mesenchymal stem cell manufacturing for multiple clinical trials in the areas of cardiology, pulmonology, dermatology, diabetes and spinal cord injury. Khan and her colleagues are dedicated to bringing regenerative medicine research to new levels by incorporating bioreactor technology in producing these mesenchymal stem cells on a large commercial scale.
Her focus is on understanding the mechanism of induced pluripotent stem cells (iPSCs) for cardiac and neurological diseases. These cells show enormous promise for regenerative medicine, which requires further exploration in the area of characterization. Khan has presented at the Food and Drug Administration’s Cell Therapy/FDA Liaison Meeting regarding characterization of clinical grade iPSCs, which could have an influence on FDA guidelines for the use of iPSC cell banks. ISCI plans to place major focus on developing new regenerative medicine products for tissue replacement and creating bioartificial organs.
CURING PARALYSIS
There are no easy problems in biomedical science, and paralysis and neurodegenerative disorders are among the most difficult. The complexity of the nervous system includes diverse cell types from neurons to astrocytes, as well as microglia, Schwann cells and oligodendrocytes. Researchers at The Miami Project to Cure Paralysis at the Miller School are trying to understand how these cells work together to develop regenerative strategies to improve function and quality of life.
“Over the years our programs have developed into a highly collaborative, multidisciplinary network with more than 200 investigators working on different aspects of central nervous system injury and repair,” said W. Dalton Dietrich, Ph.D., The Miami Project’s scientific director and senior associate dean for discovery science. “We work on spinal cord and brain injuries, nerve regeneration and, more recently, neurodegenerative disorders. We are developing novel cell therapies that are being combined with other regenerative strategies for clinical trials that are making a real difference in people’s lives.”
In recent studies, neurosurgical clinician-scientists have investigated whether pluripotent stem cells can regenerate damaged spinal cord tissue, or whether mesenchymal stem cells can treat paralysis to help patients regain motor function.
However, not all cell therapies are stem cell therapies. Scientists at The Miami Project have pioneered the use of Schwann cells as a strategy for repairing the injured spinal cord. Working in collaboration with the cell production lab at ISCI, this multidisciplinary group has developed the capacity for successfully transplanting large numbers of clinical-grade Schwann cells to produce myelin, a membrane material that insulates axons to support neuron survival and the speed of communication. Understanding the biology of these supportive cells could make replacement therapies more effective.
The Miami Project scientists are also developing new approaches for enhancing the regenerative capacity of stem and Schwann cell transplants to make them more effective. Gene therapy and testing of novel biomaterials in which cells can be successfully transplanted are enhancing cell transplant survival and their integration into host tissues for improved regeneration and recovery.
Whole-body approaches, including rehabilitation strategies, are also being combined with cell therapies to enhance the benefits of these regenerative approaches. Participants undergo months of rehabilitation before and after transplantation to synergize cell therapies with exercise. An exciting development in the regeneration field has been the appreciation of activity-based therapies to enhance functional recoveries after brain and spinal cord injury. Researchers at The Miami Project are also using neurostimulation approaches to enhance regeneration by harnessing new technologies developed by UM scientists and provided by industry partners.
In parallel, drug discovery programs focusing on axonal regeneration are showing great promise in identifying candidate compounds or genes that significantly enhance spinal cord axon regeneration. Future studies will continue to clarify what specific combination approaches will be most beneficial in regenerating the injured nervous system and promoting clinically relevant functional recovery.
There are six active FDA trials at The Miami Project, and many basic and translational projects to test new approaches to induce axonal regeneration and improve circuit function.
“What’s special at The Miami Project is that we have highly collaborative groups working together to achieve our major goals,” Dr. Dietrich said.
Investigators, including cell and molecular biologists, neuroscientists, electrophysiologists, biomedical engineers and clinicians from multiple departments and centers throughout UM, are combining their expertise to improve outcomes for patients living with these neurological disorders.

THE ALZHEIMER’S BLACK HOLE
Twenty years ago, researchers thought they had a good handle on Alzheimer’s disease. Aberrant amyloid beta proteins form layers of toxic plaques in the brain. Scientists thought if they could keep this plaque from forming, or eliminate it, they could treat the disease.
Now, however, the scientific community is less certain. A series of promising phase 3 clinical trials were mostly complete failures, producing a therapeutic black hole.
As a result, finding effective Alzheimer’s therapies has become an urgent mission. In addition to generating tremendous suffering in both patients and caregivers, this year Alzheimer’s and other dementias will cost the nation $290 billion. By 2050, as the population continues to age, the price tag could be as high as $1.1 trillion.
With the potential to regenerate damaged tissue and possibly even restore dying brain cells, stem cells may offer a solution. Barry Baumel, M.D. ’74, assistant professor of neurology, is working with Dr. Hare and others to test whether mesenchymal stem cells could provide some therapeutic benefit.
“We believe infusions of these types of stem cells have the potential to be beneficial to individuals with Alzheimer’s disease,” said Dr. Baumel, whose project is being conducted using the Diabetes Research Institute/Cell Transplant Center cGMP Umbilical Cord MSC Master Cell Bank.
Drs. Baumel and Hare were the first to infuse mesenchymal stem cells into Alzheimer’s patients. That clinical trial, which launched in 2016, was designed to test safety. The next step will be to determine if these cells can provide therapeutic benefit. The study Dr. Baumel has just begun will be the Western world’s first to test the effect of repeated injections of mesenchymal stem cells into Alzheimer’s patients over the course of one year.
Mesenchymal stem cells could reduce inflammation, as they’ve done in other tissues, and may also encourage the brain’s hippocampus to produce new neurons. They could provide a potent therapy, and the first source of hope for patients and families.
“By targeting the two antigens, we may reduce the risk of relapse. It’s like using two antibiotics in tandem against a serious infection.”
— Krishna V. Komanduri, M.D.
THE EVOLUTION OF BLOOD CANCER TREATMENTS
Though regenerative medicine is considered a relatively new discipline, cell therapies have been around for decades. Many patients with aggressive blood cancers, such as lymphomas, have received bone marrow transplants, another form of mesenchymal stem cell transplant.
In those cases, patients receive massive chemotherapy to knock out their cancer. Unfortunately, the treatments also destroy the patients’ bone marrow, which makes both red and white blood cells. Transplants were designed to restore that function.
“Initially, we thought the high-dose chemo was doing all the work and the stem cells were there to reseed the garden,” said Krishna V. Komanduri, M.D., who runs the Division of Transplantation and Cellular Therapy at Sylvester Comprehensive Cancer Center.
That understanding turned out to be incomplete. One potential complication of allogeneic stem cell transplants, in which surgeons use donor cells, is a condition called graft versus host disease (GvH), in which donor immune cells attack a patient’s normal tissues. To counter GvH, clinicians removed T cells from the transplants, but that presented an even greater problem: Patients started relapsing at higher rates.
“That led us to the notion that the immune components in these allogeneic transplants were part of the anti-cancer therapy and not just there for the ride,” Dr. Komanduri said.
Fast forward three decades, and those insights helped inspire an immunotherapy revolution — CAR-T therapy. A patient’s T cells — a type of lymphocyte that develops in the thymus gland and plays a central role in the immune response — are collected from the blood, then modified to produce special structures called chimeric antigen receptors (CARs) on their surface. This allows them to latch on to CD19 receptors, which are common on blood cancer cells.
For many patients suffering from intractable blood cancers, CAR-T has been a game-changer. In some cases, cure rates have gone from 10% to 40%. Sylvester has played a big role in this revolution, hosting some of the largest CAR-T clinical trials.
Now, Dr. Komanduri and colleagues are working to expand CAR-T, testing it on adult acute lymphoid leukemia and other blood cancers. They are also looking at other T cell therapies for liver, lung and gynecologic cancers.
But perhaps the most interesting trial, starting this fall, will test CAR-T cells that target two receptors: CD19 and CD22. Cancer cells can evolve to delete their CD19 receptor, eluding treatment. Adding a second target helps ensure they can’t hide.
“By targeting the two antigens, we may reduce the risk of relapse,” Dr. Komanduri said. “It’s like using two antibiotics in tandem against a serious infection.”
“We saw dramatic responses in some patients. … We are probably on the cusp of a revolution where we get very close to bioengineered skin.”
— Carl I. Schulman, M.D., M.S.P.H., Ph.D.
A CURE FOR SICKLE CELL?
Sickle cell is a horrific and painful disease. Mutations to hemoglobin — the protein in blood that carries oxygen — generate abnormal cell shapes, impairing blood flow.
“For people with sickle cell disease, when the hemoglobin molecules release the oxygen, they attach to each other, forming long chains that look like needles. This chain formation, called polymerization, deforms the shape of red blood cells,” explained Ofelia Alvarez, M.D., professor of clinical pediatrics and director of the Pediatric Sickle Cell Program.
Because sickle cell disease affects the blood, it affects every part of the body. In addition to causing frequent, unexpected pain, the disease is associated with stroke, pulmonary hypertension, kidney disease, leg ulcers and other complications.
Bone marrow stem cell transplants offer the possibility of a cure. Earlier this year, the Miller School was the first in South Florida to perform the procedure on an adult sickle cell patient, a 31-year-old woman who no longer must endure agonizing sickle cell crises.
Unfortunately, the treatment is not widely available. The sticking point is finding donors who are good matches.
“A patient must have a sibling match,” said Thomas Harrington, M.D. ’85, who directs the Adult Sickle Cell Program at UM/Jackson Memorial Hospital. “But often brothers and sisters have the same disease, or the patient may not have a full sibling. Unrelated donor and haploidentical transplants (from a parent) are areas of study and have been done in children.”
But the Miller School is poised to open a South Florida study site for a promising clinical study sponsored by Bluebird Bio. Rather than relying on donor stem cells, the trial uses the patient’s own cells, removing them and editing the defective DNA to insert a functional hemoglobin gene. The patient is then treated with chemotherapy to destroy their diseased marrow and the new cells are infused. Drs. Alvarez and Harrington are working together to bring the trial to Miami.
“It’s potentially a cure,” Dr. Harrington said, “but it will take a while to complete all the work to validate the treatment’s safety.”
NEXT-GENERATION BURN CARE
Treatments for third-degree burns have changed little over the past 50 years, but the promise of stem cells has renewed hope for a breakthrough.
“Surgeons cut off the burned skin and replace it with skin grafts from unaffected places on the patient’s body,” explained Carl I. Schulman, M.D., M.S.P.H. ’04, Ph.D. ’11, professor of surgery and executive dean for research. “But the grafting process is painful and leaves scars.”
Dr. Schulman is working with Evangelos Badiavas, M.D., Ph.D., professor of dermatology, to find better solutions. Dr. Badiavas’ previous work at ISCI had demonstrated that longstanding non-healing wounds in patients (some of 10 years or more duration) could be healed with the application of bone marrow stem cells. In a new study, patients with deep second-degree burns were treated with allogeneic mesenchymal stem cells. The study was primarily designed to assess safety, but it also produced many encouraging results.
“We saw dramatic responses in some patients,” Dr. Schulman said. “Their wounds healed faster and better than expected.”
The study was funded by the U.S. Department of Defense to improve battlefield burn treatments. Mesenchymal stem cells are ideal, Dr. Schulman said, because they can be deployed anywhere. “Mesenchymal stem cells are both anti-inflammatory and provide cellular signaling that tells wounds to heal more like normal skin and less like scarred skin.”
The ultimate goal is lab-grown replacement tissue, though that is a technically challenging proposition. Skin is a complex organ and contains many cell types. Putting them all together is going to take time, but it’s coming.
“We are probably on the cusp of a revolution,” Dr. Schulman said, “where we get very close to bioengineered skin.”

TYPE 1 DIABETES: THE TWO-PART CHALLENGE
Once called juvenile diabetes, type 1 diabetes is an autoimmune disease, in which the body turns against itself and immune cells begin to destroy the pancreatic islet cells that monitor blood glucose levels and release insulin to tell cells to take in that glucose. Cells get energy, and the body maintains healthy blood sugar levels.
Islet cells work with exquisite precision, which humans have a hard time replicating. Though injected insulin converts this deadly disease into a chronic one, it’s difficult for even the most careful diabetes patient to mimic the natural function of the pancreas.
Overabundant glucose is toxic, damaging blood vessels, kidneys, the eyes, and the heart. Transplanted islet cells could potentially cure the disease, but it’s a two-part challenge: First, surgeons have to successfully engraft insulin-producing cells, then they have to prevent the autoimmune response from killing them all over again.
“How do we manipulate the immune system to prevent rejection or recurrent autoimmunity, but do it without making the patient more prone to infection and cancer?” asked Norma Sue Kenyon, Ph.D., the Martin Kleiman Professor of Surgery, Microbiology and Immunology, and Biomedical Engineering.
Dr. Kenyon is working with Camillo Ricordi, M.D., who directs the Miller School’s renowned Diabetes Research Institute (DRI), and others to bring more effective islet cell transplants to patients. DRI researchers have successfully coaxed pluripotent stem cells into becoming transplant-ready islet cells, which could alleviate the cell shortage.
“There are a thousand people who die each year from lack of suitable donor cells,” Dr. Ricordi said. “We are now moving toward stem cell-derived, insulin-producing cells, which will provide an unlimited supply.”
Simultaneously, Miller School scientists are pursuing several strategies to control the immune response. Dr. Ricordi and others conducted an eight-year trial, infusing mesenchymal stem cells to increase insulin production and reduce the symptoms associated with diabetes. The protocol cut those complications in half.
Dr. Kenyon hopes to augment these protective traits with more selective immune system inhibition. She noted that 20 years ago a drug was tested that successfully targeted CD40 ligand, a molecule on activated T cells that governs their ability to mediate an immune response. The drug was tremendously effective at preventing immune rejection but caused unacceptable side effects.
Now, Miller School researchers are working with a company to develop a similar drug, but without the disqualifying side effects. Combining it with mesenchymal stem cells could make islet transplants more viable.
Another line of research is looking at regulatory T cells, or Tregs. The immune system is exquisitely balanced between cells that potentially attack cancer, pathogens and even your own tissues and other cells that regulate those attacks. Effector and autoreactive T cells destroy invaders and tissues; Tregs tell them to stop.
Thomas Malek, Ph.D., professor and chair of the Department of Microbiology and Immunology, says these interactions offer a window to more precisely control the immune system.
“A lot of cell therapies and regenerative medicine techniques lead to unwanted immune responses, which must be controlled,” Dr. Malek said. “We need to prevent the immune system from attacking transplanted cells and one’s own tissue in the case of autoimmune disease.”
Dr. Malek has spent nearly 40 years studying a molecule called IL2. At first, scientists thought IL2 only boosted effector T cells, but years of research have generated a more complex picture.
“In some ways, IL2 is like a rheostat,” Dr. Malek said. “At certain levels, it induces immunity. At lower levels, it targets regulatory T cells.”
In other words, IL2 acts as both an immune system stimulant and a brake, depending on the dosage. Tregs have more IL2 receptors, which makes them more sensitive to lower doses than effector T cells. Dr. Malek hopes to leverage this gap to encourage Tregs to prevent the immune system from attacking islet cell transplants and self-tissues without suppressing immunity that makes patients become susceptible to cancer or opportunistic infections.
“We are working on a clinical trial in type 1 diabetes to address the autoimmune attack,” he said. “But this research could be applied to inflammatory conditions, autoimmune disease and other kinds of transplants.”
The Potential Power
of Cell Therapy
Miller School scientists are working to solve many of the world’s health problems by harnessing cellular therapy to repair or replace diseased tissue in the body.
“We are working on a clinical trial in type 1 diabetes … But this research could be applied to inflammatory conditions, autoimmune disease and other kinds of transplants.”
— Thomas Malek, Ph.D.
FUTURE DIRECTIONS
The cell production labs at ISCI and DRI are collaborating broadly across the campus with many departments to advance other therapeutic concepts that one day may be added to the regenerative medicine armamentarium.
One approach is to develop cellular constituents, such as microscopic vesicles known as extracellular vesicles or exosomes. These smaller particles may have therapeutic value and are being developed by Aisha Khan at ISCI and other Miller School researchers for a variety of conditions.
More ambitious, but on the drawing board, is research to generate an entire organ using cell therapy. Many collaborators across campus are working on using induced pluripotent stem cells to generate entire organs, such as hearts or kidneys. Khan, Dr. Hare and Dr. Chatzistergos are leading multiple projects funded by the National Heart, Lung and Blood Institute to develop these cells for cardiac and other conditions. While still a long way off, this approach could potentially revolutionize solid organ transplantation.
GETTING TREATMENTS TO PATIENTS
Developing stem cell-based treatments is a slow, painstaking process, and it’s difficult to tell patients they have to wait. But that pace is intentional, because it keeps poorly understood therapies from causing harm.
Unfortunately, opportunistic stem cell “clinics” have been set up around the world, offering unproven therapies. Recently, the Food and Drug Administration shut down two such facilities in South Florida.
“We’ve tried to be the counterinfluence to these clinics by insisting that cell products are drugs and should be administered as drugs,” Dr. Hare said. “There’s no role for clinics that make false promises.”
Many scientists and physicians note that these fly-by-night operations give regenerative medicine a bad reputation, but say they are also motivated to work harder to bring proven therapies to patients as rapidly as possible.
“Many patients go out of the country and pay a lot of money for these untested therapies,” Dr. Ivonne Schulman said. “Nobody really knows if they work, or if they don’t work, if they’re dangerous. We are conducting the rigorous clinical research first. If the treatments work — wonderful. If not, we figure out why and try again.”
Clinical trials move promising research from the scientists’ bench into patient studies, but at some point, life sciences companies must get involved. To speed that process, the University of Miami is actively licensing cell therapy technologies to biotech and pharma companies. UM’s U Innovation program is working with a long list of companies, including Longeveron, a startup launched by Dr. Hare that’s looking at cell-based solutions to address healthy aging; Aegle Therapeutics, a company launched by Dr. Badiavas that focuses on severe dermatological conditions; and Aceso Therapeutics, a company launched by a group led by Dr. Dietrich that is dedicated to developing Schwann cell therapies for a number of neurological disorders.
“One of our goals, wherever we can,” Dr. Kenyon said, “is to license our discoveries to start-ups or established companies. The more we do that, the more we can help patients.”![]()